When Life-Saving Antibodies Turn Against Red Blood Cells: Understanding IVIG-Induced Hemolysis
A single bottle of IVIG contains antibodies from thousands of blood donors. These antibodies can help regulate the immune system and save lives, but sometimes they may mistakenly attack a patient’s own red blood cells. This shows why knowing about blood groups and antibody levels is so important in medicine.
2/18/20264 min read
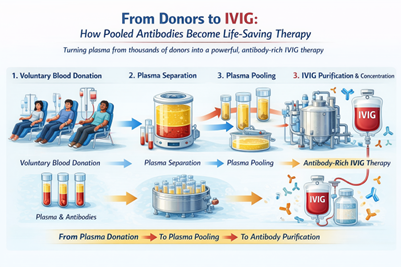
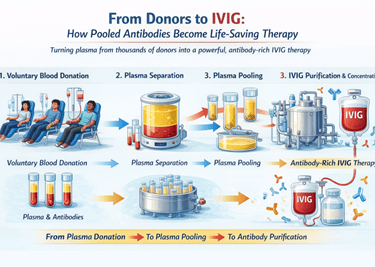
In this blog, we explore how a life-saving therapy derived from donated plasma can occasionally lead to an unexpected complication.
You’ll learn:
• What IVIG therapy is and how antibodies from thousands of donors are pooled together to create it
• How anti-A and anti-B antibodies in IVIG can sometimes trigger hemolysis in patients
• Why antibody titer monitoring and ABO compatibility are critical for patient safety
• How improved diagnostic tools and rapid blood typing technologies could help reduce transfusion-related risks.
Modern medicine relies heavily on treatments made from donated human blood. One important example is Intravenous Immunoglobulin (IVIG), which is used to treat many immune-related diseases. From autoimmune disorders to neurological problems and immune deficiencies, IVIG has improved the lives of thousands of patients around the world.
However, like many strong treatments, IVIG does carry some risks. In rare cases, patients may develop hemolysis, where their red blood cells break down too quickly. This condition, called IVIG-induced hemolysis, reminds us how delicate the balance is between antibodies, blood groups, and immune responses (Sandler & Mallory, 2017). It also shows why careful monitoring and accurate blood tests are so important in healthcare.
Understanding IVIG Therapy
IVIG is a plasma-derived medicine that contains immunoglobulin G (IgG) antibodies pooled from thousands of healthy blood donor (Berger, 2011). Because the antibodies come from such a diverse donor population, the resulting therapy contains a wide variety of immune molecules capable of recognizing and neutralizing pathogens or modulating abnormal immune responses.For patients whose immune systems are compromised or dysregulated, IVIG can be life-saving.
It is commonly used to treat conditions such as:
● primary immunodeficiency disorders
● autoimmune diseases
● Kawasaki disease
● and certain neurological syndromes.
In many cases, IVIG works by calming excessive immune activity or by providing antibodies that the patient’s own immune system cannot produce effectively.
While IVIG is usually safe, it comes from pooled plasma, so it contains natural antibodies against blood group antigens, especially anti-A and anti-B antibodies. Most of the time, these antibodies are too low to cause problems. But in some cases, especially with high doses of IVIG, they can cause unexpected reactions in the bloodstream.
When Antibodies Target Red Blood Cells
Hemolysis occurs when red blood cells are destroyed faster than the body can replace them. In the context of IVIG therapy, this process typically begins when antibodies present in the IVIG preparation bind to blood group antigens on the patient’s red blood cells.
For example, if a patient with blood group A, B, or AB receives IVIG containing antibodies against those groups, the antibodies may attach to their red blood cells. Once coated, the immune system can identify and remove these cells, which lowers hemoglobin levels. This issue is rare but happens more often in patients getting high doses of IVIG, especially those with non-O blood groups. Inflammation can increase this risk because the immune system is already active (Kessary-Shoham et al., 1999).
Symptoms may appear a few days after the infusion and include tiredness, jaundice, dark urine, and a noticeable drop in hemoglobin levels. In severe cases, patients might need extra care or blood transfusions to help stabilize them.
Understanding Antibody Levels
A key factor in the risk of IVIG-induced hemolysis is the antibody titre in the IVIG product. Antibody titre means the concentration or strength of antibodies that can react with red blood cell antigens.
Because IVIG is made from plasma collected from many donors, antibody levels can differ between batches. Some donors naturally have higher levels of anti-A or anti-B antibodies, and when plasma from many donors is mixed, these antibodies can build up to higher levels.
When IVIG batches have high levels of these antibodies, the risk of hemolysis in vulnerable patients goes up. That’s why monitoring antibody titres is important to keep plasma-derived therapies safe.
Improving Safety Through Advanced Testing
To minimize these risks, manufacturers have increasingly adopted improved quality control methods. One promising approach is the glass bead-based antibody titre assay, which has been proposed as a reliable method for measuring anti-A and anti-B antibody levels during IVIG production. In this method, antigen-coated glass beads bind antibodies in a sample. By observing how strongly these antibodies bind to the beads, manufacturers can estimate antibody titres more accurately. Such assays allow potentially high-titre batches to be identified and managed before the product reaches patients. These kinds of quality assurance checks play an essential role in ensuring that therapies derived from donated plasma remain both effective and safe for clinical use.
Why Blood Donation and Blood Typing Still Matter
At its core, the story of IVIG-induced hemolysis starts long before the therapy reaches a patient. It begins with blood donation.
Plasma used to manufacture IVIG originates from thousands of volunteer donors. Each donor has a unique immunological profile, including naturally occurring antibodies associated with their blood group. Some individuals naturally possess higher levels of anti-A or anti-B antibodies, which can influence the antibody composition of pooled plasma products.
That’s why knowing donor blood groups and antibody levels is key to keeping transfusion medicine and plasma therapies safe. Careful donor screening, accurate blood typing, and strong antibody testing help make sure life-saving treatments don’t bring new risks.
Knowing ABO blood group compatibility is still very important in clinical care. Understanding both the patient’s blood group and the antibodies in treatments helps doctors predict and handle possible problems. Accurate blood typing, antibody monitoring, and better diagnostic tools are the foundation of safer transfusions and plasma therapies. This shows that blood typing science goes well beyond just transfusions.


Looking Ahead: The Need for Faster and Smarter Blood Testing
As healthcare grows and more blood-derived therapies are needed, having fast, reliable, and local diagnostic tools becomes even more important.
Traditional lab tests, though accurate, can take time, cost a lot, and rely on central labs. In critical care, delays in blood typing or antibody screening can raise risks during transfusions or treatments.
Developing rapid, cost-effective, and decentralized assays for blood group determination and antibody detection could significantly improve patient safety. Faster testing would enable clinicians to make informed decisions more quickly, reduce the likelihood of transfusion-related complications, and ensure that compatible blood products are available when they are needed most. Beyond clinical settings, such innovations could also strengthen blood donation networks, improve donor screening processes, and enhance quality control in plasma-derived therapies. (Davenport, 2015)
